
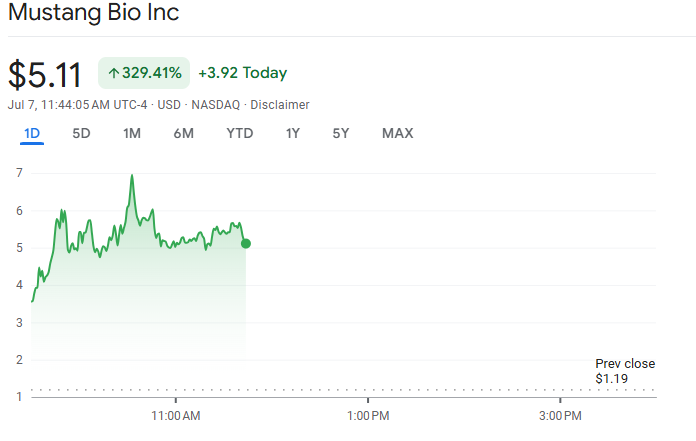
Shares of Mustang Bio (NASDAQ: MBIO) surged on Monday as investors responded to a significant approval from the Food and Drug Administration (FDA).
By press time, Mustang Bio’s stock was trading at $5.11, marking a 329% surge. Over the past week, the equity has rallied by more than 400%.
Notably, the stock is part of the portfolio of the world’s largest investment management firm, BlackRock (NYSE: BLK), which owns 1,665 shares in the company.
The rally followed news that the FDA granted orphan drug designation to Mustang’s experimental treatment MB-101, designed to combat rare and aggressive forms of brain cancer.
The FDA’s decision covers MB-101 as a therapy for recurrent diffuse and anaplastic astrocytoma and glioblastoma, challenging brain cancers with limited treatment options.
Notably, orphan drug designation offers significant advantages, including tax credits for clinical development and seven years of market exclusivity upon securing final approval. Notably, the FDA’s green light applies to a broader patient group than Mustang had originally proposed.
MB-101 is a CAR T-cell therapy targeting IL13Ra2, which is overexpressed in aggressive brain tumors. In a Phase 1 trial, 50% of patients achieved disease stabilization or better, with two experiencing complete responses lasting 7.5 months or longer, linked to high intratumoral CD3+ T-cell levels.
Mustang is also developing MB-109, a combination of MB-101 and MB-108, an oncolytic virus therapy. Progress depends on securing additional funding or partnerships. Both treatments are in Phase 1 trials at City of Hope and the University of Alabama at Birmingham.
Relief for Mustang amid financial woes
Generally, the approval comes as a boost for the company, which only in February disclosed that it may need to seek bankruptcy protection or pursue other options that could wipe out shareholder value if it cannot raise additional capital.
As of the end of September 2024, the company reported $3.5 million in cash and cash equivalents, with an accumulated deficit of $395.8 million.
Mustang develops cell therapies for hard-to-treat cancers, focusing on CAR T treatments licensed from research institutions. Its business model relies on partnering for preclinical and clinical development, then transferring the technology in-house or to a contract manufacturer to run its trials.
Featured image via Shutterstock
You may also like
Archives
- April 2026
- March 2026
- February 2026
- January 2026
- December 2025
- November 2025
- October 2025
- September 2025
- August 2025
- July 2025
- June 2025
- May 2025
- April 2025
- March 2025
- February 2025
- January 2025
- December 2024
- November 2024
- October 2024
- September 2024
- January 2024
- December 2023
- January 2023
- December 2022
- January 2022
- December 2021
- January 2021
- December 2020
- December 2019